About 42% of U.S. adults meet the clinical definition of obesity, a widespread health issue that influences treatment choices for diabetes and weight management. Choosing the right medication dose affects long-term health, energy, and safety.
- Correct medication dosing is essential to maximize benefits like blood sugar and weight control while minimizing side effects.
- Work with a licensed healthcare provider or clinic to develop a personalized treatment plan and receive appropriate monitoring.
Starting a new prescription or weight-management plan can feel overwhelming. Identifying the optimal tirzepatide dose, one that balances efficacy for blood glucose and weight reduction with tolerability- is central to success. Individual factors such as age, kidney function, other medications, and treatment goals make precision important.
This guide explains how tirzepatide works, the common dosing schedules, practical administration tips, safety considerations, and next steps to discuss with your clinician so you can pursue safe, effective weight loss and diabetes care.
Understanding Tirzepatide and Why Proper Dosing Matters
Choosing the correct tirzepatide dosage is central to achieving treatment goals while minimizing adverse effects. Tirzepatide is a novel dual incretin receptor agonist that targets both gastric inhibitory polypeptide (GIP) and glucagon‑like peptide‑1 (GLP‑1) pathways, producing benefits for blood glucose control and body weight.
What Is Tirzepatide and How Does It Work
Tirzepatide acts on two receptor systems that regulate metabolism. By activating GIP and GLP‑1 receptors, the drug improves glycemic control and reduces appetite through several complementary actions:
Here’s how tirzepatide works:
- Augments insulin secretion when blood glucose is elevated, aiding glycemic control.
- Suppresses inappropriate glucagon release, which helps lower blood sugar.
- Slows gastric emptying, which can reduce postprandial glucose spikes and curb appetite.
- Reduces appetite and caloric intake, contributing to weight loss over time.
The Critical Role of Correct Dosing
Accurate dosing optimizes the balance between efficacy (improved glycemic control and weight loss) and tolerability (reduced gastrointestinal and other side effects). A gradual, evidence-based titration minimizes nausea and vomiting while allowing patients to reach effective maintenance doses.
Benefits of the right tirzepatide dose include:
- Improved blood glucose and HbA1c control when used for type 2 diabetes.
- Support for clinically meaningful weight loss in many patients.
- Lower incidence and severity of dose‑related side effects when titrated appropriately.
- Greater likelihood that patients will continue and adhere to their treatment plan.
Who Should Consider Tirzepatide Treatment
Currently, tirzepatide is prescribed primarily for adults with type 2 diabetes as part of a comprehensive treatment plan; clinicians also evaluate its role in weight management where appropriate. Deciding to start tirzepatide should be a shared decision between you and a licensed clinician, based on individual health factors.
Factors clinicians consider when evaluating tirzepatide use include:
- Diagnosis of type 2 diabetes and current glycemic control.
- Previous response to other diabetes or weight‑loss medications.
- Comorbid conditions such as renal impairment or a history of pancreatitis.
- Concomitant medications that increase hypoglycemia risk (for example, insulin or sulfonylureas).
Standard Tirzepatide Dosage Guidelines and Protocols
Adhering to the manufacturer and FDA‑approved titration schedule for tirzepatide helps balance effectiveness with tolerability. Tirzepatide is administered as a once‑weekly subcutaneous injection; the recommended escalation is designed to limit gastrointestinal side effects while moving patients to an effective maintenance dose.
FDA-Approved Tirzepatide Dosing Schedule
The labeled titration schedule provides stepwise dose increases over several weeks so clinicians and patients can monitor response and side effects.
The labeled titration schedule provides stepwise dose increases over several weeks so clinicians and patients can monitor response and side effects.
| Week | Tirzepatide Dose |
| 1-4 | 2.5 mg once weekly |
| 5-8 | 5 mg once weekly |
| 9-12 | 7.5 mg or 10 mg once weekly (based on tolerance) |
| After 12 weeks | Maintenance dose: typically 5 mg, 7.5 mg, 10 mg (and higher doses may be considered per current labeling and clinician judgment) |
Recommended Starting Dose
Begin with 2.5 mg once weekly for at least four weeks. This initial low dose helps the body adjust and reduces the risk of dose‑related gastrointestinal effects.
Maintenance Dose Ranges
After the initial period, clinicians commonly increase to 5 mg once weekly and may escalate to 7.5 mg or 10 mg depending on clinical response and tolerability. The choice of maintenance dose reflects treatment goals (glycemic control vs. body weight reduction), side‑effect profile, and individual patient factors.
Maximum Recommended Tirzepatide Amount
The maximum recommended dose in the product labeling should be confirmed against the latest prescribing information; treatment decisions about higher doses require clinician review of benefits versus risks. Follow your prescriber’s guidance regarding the upper dose for your situation.
Why this titration matters: slow stepwise increases reduce nausea and vomiting risk and let clinicians assess glucose, weight, and overall tolerance at each dose. Discuss titration goals, monitoring plans (for example, frequency of HbA1c checks and weight assessments), and any adjustments with your licensed healthcare provider before changing doses.
Tirzepatide Administration: Step-by-Step Instructions
Proper administration of tirzepatide ensures the prescribed dose delivers intended benefits for blood glucose and weight while minimizing local and systemic side effects. Tirzepatide is given as a once‑weekly subcutaneous injection; learning correct technique and storage makes treatment safer and more effective.
How to Take Tirzepatide Injections
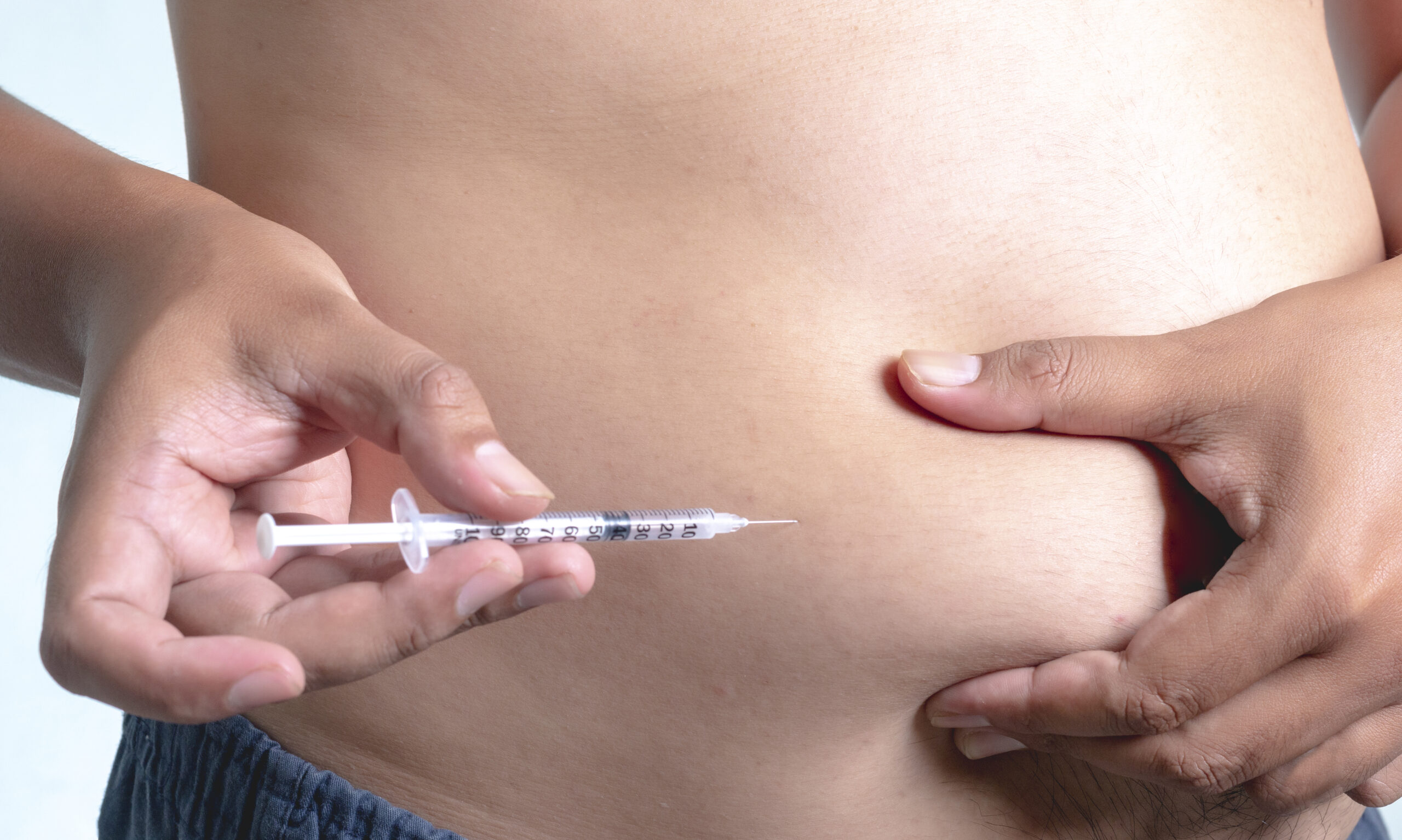
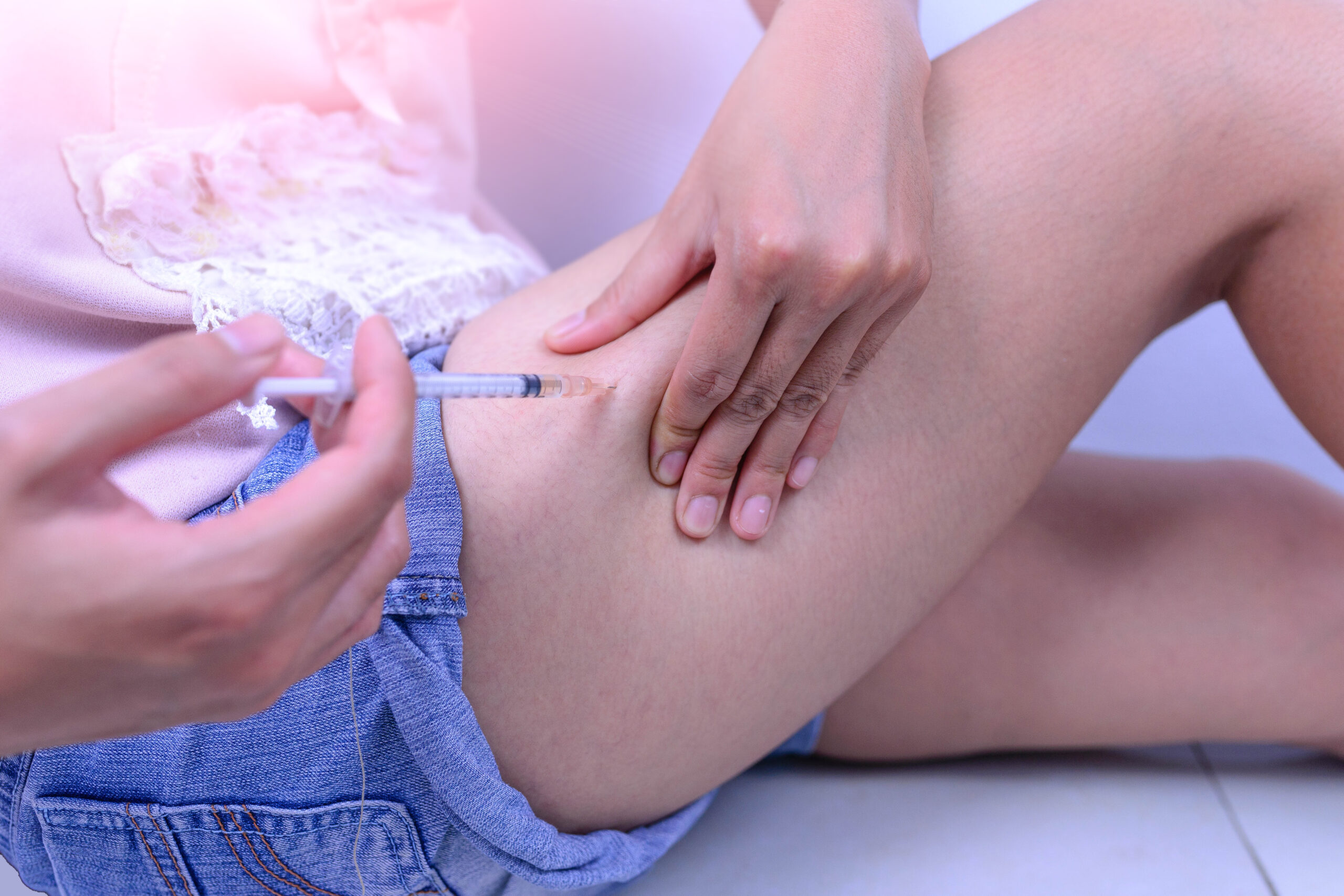
Patients typically self-administer tirzepatide once weekly. The medication is injected under the skin (subcutaneously), commonly into the abdomen, thigh, or upper arm.
General steps for a subcutaneous injection (follow the device instructions provided with your prescription and confirm technique with your clinician):
- Wash your hands and prepare a clean, well‑lit workspace.
- Inspect the injection pen or syringe and medication for damage or discoloration.
- Choose an appropriate injection site (see rotation guidance below) and clean the area with an alcohol swab if instructed.
- For most adults, insert the needle at a 90° angle to the skin; follow the manufacturer’s device directions if they recommend a different angle or a skin‑fold technique for thin individuals.
- Press to deliver the full dose, then keep the needle in place for the number of seconds recommended by the device instructions before withdrawing.
- Dispose of needles or pens in an approved sharps container and store the pen per instructions.
Proper Injection Sites and Rotation Technique
Rotate injection sites to reduce the risk of lipohypertrophy (localized fat accumulation) and injection site reactions. Avoid injecting into areas with redness, scarring, bruises, or broken skin.
| Injection Site | Benefits | Precautions |
| Abdomen | Easy self‑access; consistent absorption | Avoid within 2 inches (5 cm) of the navel; rotate locations around the abdomen |
| Thigh | Large surface area; convenient for many people | Rotate between different sections of the thigh; avoid areas with scars or visible veins |
| Upper Arm | Alternative site; useful if abdominal or thigh sites are problematic | May be harder to self‑administer; rotate within the upper arm areas |
Timing and What to Do If You Miss a Dose
Pick a day of the week that’s easy to remember and take tirzepatide on that same day each week. Consistency supports adherence and steady therapeutic levels.
If you miss a scheduled weekly injection, follow the most current prescribing information or your clinician’s advice. In many cases, manufacturers recommend taking a missed weekly dose as soon as you remember within a specified window (check the product label for exact timing). Do not take two doses too close together—if the recommended window has passed, skip the missed dose and resume your regular weekly schedule. Confirm the exact missed‑dose instructions with your prescriber or pharmacist.
Storage Requirements and Medication Handling
Proper storage preserves medication potency. Follow the product insert, but common recommendations include:
- Store unopened pens in the refrigerator between 36°F and 46°F (2°C and 8°C).
- Do not freeze tirzepatide; throw away if it has been frozen.
- If the product instructions allow, an in‑use pen may be kept at room temperature for a limited number of days—check the label for the exact duration and temperature limits.
- Keep pens in their original carton to protect from light when specified.
- Always inspect the medication before use; if you notice particles, discoloration, or damage, do not use and contact your pharmacy or clinician.
Want help learning injections? Ask your clinician, diabetes educator, or pharmacist for a demonstration and to review proper sharps disposal. They can also confirm device‑specific steps and whether a skin‑fold or 90° technique is appropriate for you.
Dose Titration and Finding Your Optimal Tirzepatide Dose
Finding the right tirzepatide dose is an individualized process that balances therapeutic goals (improved glycemic control and weight loss) against tolerability. Dose titration helps most patients reach an effective maintenance dose while reducing the chance of dose‑related side effects.
Understanding the Dose Escalation Process
Titration follows a stepwise plan: start low, monitor response and side effects, then increase doses at planned intervals if tolerated. This gradual escalation reduces nausea and vomiting and allows clinicians to evaluate the benefit at each dose.
Typical escalation steps (see your prescriber for label‑specific details):
- Begin with the starting dose (for example, 2.5 mg once weekly).
- Monitor symptoms and early effects on appetite and blood glucose.
- If tolerated, increase to the next dose after the recommended period (commonly every 4 weeks) until a maintenance dose is reached.
- Reassess goals and tolerability regularly and adjust the plan as needed.
When and How to Increase Your Dosage
Decisions to increase dose are based on clinical response and tolerability. Your clinician will typically reassess you at scheduled follow‑up visits (for example, at 4‑week intervals during titration) to evaluate:
- Glycemic control (home glucose readings and, over time, HbA1c).
- Side effects—particularly gastrointestinal symptoms that are dose‑related.
- Weight trajectory and goals for body weight or weight loss.
If you are taking insulin or sulfonylureas, your clinician may proactively adjust those medications to reduce hypoglycemia risk when increasing tirzepatide doses.
Individual Factors Affecting Dose Optimization
Several patient factors alter the optimal dose or monitoring approach. Discuss these with your prescriber:
- Renal function: patients with renal impairment may need closer monitoring of adverse effects and renal parameters.
- Age: older adults may be more susceptible to side effects and falls related to hypoglycemia—individualize dosing and monitoring.
- Concomitant medications: drugs that increase hypoglycemia risk (insulin, sulfonylureas) or whose absorption may be affected by slower gastric emptying require attention.
- Baseline weight and treatment goals: dose selection may differ if primary goal is aggressive weight loss versus modest glycemic improvement.
Monitoring Progress and Adjusting Treatment
Structured monitoring helps identify the optimal maintenance dose and detect adverse events early. Recommended monitoring elements (confirm exact timing with your clinician):
- Home glucose checks or continuous glucose monitoring as indicated; review patterns at follow‑up visits.
- HbA1c measurements every 3 months until stable, then per clinician guidance.
- Weight and body‑weight trend at each visit to gauge response and adjust goals.
- Assessment of GI tolerability, injection site reactions, and any new symptoms (e.g., persistent abdominal pain).
- Medication review at every visit to evaluate interactions and hypoglycemia risk.
Patient checklist for titration visits: bring recent glucose logs, a current medication list (including over‑the‑counter drugs), note any side effects or missed doses, and be prepared to discuss weight and treatment goals. Use shared decision‑making with your clinician to select a maintenance dose that aligns with your health priorities while minimizing risk.
Safety Considerations and Side Effect Management
Using tirzepatide safely requires understanding common and serious side effects, how risks change with dose, and what monitoring or medication adjustments may be needed. Work with your licensed clinician to balance benefits (glycemic control and weight loss) against potential harms and to create a plan for early recognition and management of adverse effects.
Common Side Effects at Different Dosage Levels
Tirzepatide commonly causes gastrointestinal side effects, which are generally dose‑related and tend to be most pronounced during upward titration. Typical adverse effects include:
- Nausea and vomiting- more frequent at higher doses and often improve over time with continued use and slow titration.
- Diarrhea and abdominal pain- usually transient but report persistent or severe symptoms.
- Injection site reactions- mild redness or discomfort where the medication is injected.
Quantified incidence rates vary by clinical trial and dose; refer to the product label or discuss trial data with your clinician for numeric estimates relevant to specific doses.
Warning Signs That Require Immediate Attention or Dose Adjustment
Certain symptoms should prompt urgent evaluation and may necessitate stopping tirzepatide or reducing the dose. Seek immediate care if you experience:
- Severe, persistent abdominal pain or pain that radiates to the back (possible pancreatitis).
- Vomiting blood or black, tarry stools (signs of gastrointestinal bleeding).
- Symptoms of severe dehydration, fainting, or severe weakness—especially if associated with prolonged vomiting or diarrhea.
If you develop any of these signs, stop the medication and contact your clinician or emergency services promptly.
Drug Interactions, Hypoglycemia Risk, and Contraindications
Tirzepatide can affect the absorption of orally administered medications by slowing gastric emptying; medication timing and monitoring may need adjustment for drugs with narrow therapeutic windows. Important interaction and safety considerations include:
- Hypoglycemia risk rises when tirzepatide is combined with insulin or insulin secretagogues (e.g., sulfonylureas). Your clinician may reduce the dose of these agents when starting or increasing tirzepatide to lower the risk of low blood sugar.
- Tirzepatide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) and in patients with multiple endocrine neoplasia syndrome type 2 (MEN2) per labeling; ensure your clinician reviews your personal and family history.
- Discuss all medications and supplements with your prescriber so they can assess absorption concerns and interaction risk; adjust dosing or monitoring as recommended.
Special Considerations for Patient Groups
Certain populations require individualized assessment and closer monitoring:
| Patient Group | Considerations |
| Patients with Renal Impairment | Monitor renal function and fluid status closely; adjust concomitant medications as needed and watch for dehydration from GI side effects. |
| History of Pancreatitis | Use with caution; promptly evaluate abdominal pain, and avoid if recurrent pancreatitis is suspected. |
| Pregnant or Breastfeeding Women | Limited data on safety in pregnancy or lactation—discuss risks and alternatives with a clinician before starting tirzepatide. |
When to Seek Emergency Care and Practical Safety Tips
Seek emergency care for severe abdominal pain, persistent vomiting with inability to keep fluids down, signs of GI bleeding, or severe hypoglycemia (loss of consciousness or seizure). Practical tips to reduce risk include:
- Report persistent or worsening GI symptoms early- your clinician may slow titration or adjust other medications.
- Carry supplies to treat hypoglycemia if you are on insulin or sulfonylureas and have instructions from your clinician about when to lower those agents.
- Keep a current medication list and share it at every visit so potential interactions (and effects on blood sugar) are reviewed.
- Monitor weight and any unexpected rapid weight loss or changes in appetite, and report these during follow‑up.
Discuss any concerns with your healthcare team so they can tailor monitoring (for example, checks for hypoglycemia, pancreatitis, or gallbladder disease) to your medical history and the tirzepatide dose you are using.
Accessing Tirzepatide Treatment
Obtaining tirzepatide starts with a licensed clinician who can evaluate whether the medication fits your medical needs and treatment goals. Several types of qualified providers prescribe and monitor tirzepatide, including endocrinologists, primary care physicians, diabetes clinics, and weight‑management specialists. Your pharmacist and diabetes educator can also support administration training and ongoing adherence.
Where to Seek Care and What Prescribers Will Review
When you consult a clinician about tirzepatide, they will review key elements of your health and medications to determine appropriateness and plan safe dosing:
- Medical history, including diagnosis of type 2 diabetes and prior treatments.
- Family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia type 2 (MEN2), which are contraindications.
- Current medications and supplements to assess interaction and hypoglycemia risk.
- Kidney function, liver health, pregnancy or breastfeeding status, and any history of pancreatitis or gallbladder disease.
Practical Steps Before and After Prescription
To streamline the process and support safe treatment, prepare the following before your visit:
- Bring a current medication list (including over‑the‑counter drugs and supplements) and your medical history.
- Obtain baseline labs as recommended by your clinician (for example, HbA1c, renal function, and other tests they request).
- Be ready to discuss treatment goals (glycemic control, weight loss, maintenance) and any concerns about side effects or dosing.
After receiving a prescription, arrange follow‑up visits per your clinician’s plan for titration, monitoring, and maintenance. Some insurers require prior authorization—check with your clinician’s office or pharmacy about coverage and prescription fulfillment.
Support and Follow‑Up
Ongoing monitoring and support are essential to successful treatment. Expect your prescriber to set a schedule for follow‑up visits and tests to track blood glucose, weight, and tolerability. Seek help from a diabetes educator or pharmacist for injection training, sharps disposal guidance, and counseling on how tirzepatide may affect other medications.
If you have questions about starting tirzepatide, talk with a licensed healthcare provider to determine whether it is appropriate for you and to create a personalized, safe treatment plan.
Conclusion
Choosing the right tirzepatide dose is essential to maximize benefits—improved glycemic control and weight loss—while minimizing risks and side effects. This guide reviewed how tirzepatide works, typical dosing schedules and titration strategies, administration tips, and safety considerations so you can discuss an individualized plan with your clinician.
Key takeaways:
- Start low and titrate slowly to improve tolerability and reach an effective maintenance dose.
- Monitor blood glucose, HbA1c, body weight, and any gastrointestinal symptoms during dose increases.
- Report severe abdominal pain, persistent vomiting, signs of low blood sugar, or other concerning symptoms immediately.
- Review your full medication list with your prescriber—co‑therapy with insulin or sulfonylureas may require dose adjustments to reduce hypoglycemia risk.
Before beginning or changing tirzepatide therapy, consult a licensed healthcare provider—such as an endocrinologist, primary care physician, or diabetes specialist—who can write a prescription, arrange baseline labs, and set a safe follow‑up and monitoring plan. Bring a current medication list and any recent lab results to your appointment to help the clinician tailor dosing and maintenance strategy to your needs.
With careful titration, close monitoring, and clinician support, many people experience meaningful weight loss and improved diabetes control on tirzepatide. Discuss goals, potential risks, and a follow‑up schedule with your prescriber to ensure the safest, most effective treatment plan for you.